Ocean Iron Fertilization
“Give me a half tanker of iron, and I will give you an ice age.” – John Martin
What is ocean iron fertilization?
This is a concept introduced in the 80’s by the late oceanographer John Martin. It refers to the geoengineering technology of adding iron to high-nutrient low chlorophyll (HNLC) waters.
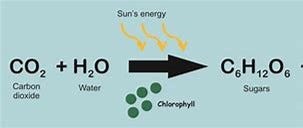
Such an addition would promote an algae bloom. Algae live and photosynthesize in surface waters of both fresh and salt environments. Because algae photosynthesize, they take in carbon dioxide (CO2), which seems to be the bad guy in climate change.
Algae take in carbon dioxide, sink to the ocean depths, taking the CO2 with them. This is one way that the ocean sequesters carbon.
Let’s go back a bit- why is carbon dioxide the bad guy?!
CO2 enters the ocean through biological, chemical, and physical interactions, such as photosynthesis (as described above) and air-sea exchange. Now, CO2 entering the ocean is natural. The ocean’s ability to sequester CO2 is one of the ways it helps to regulate the climate. However, if there is an abundance of CO2, then the effects tend to go south.
And the industrial revolution tends to get a lot of heat (haha) for the beginning of the CO2 party. Burning fossil fuels and tearing down forests pumps carbon into the atmosphere, which combines with O2 to make CO2. Since the industrial revolution, the pH of surface waters has decreased by 0.1. pH is on a logarithmic scale, (negative log of hydrogen ions in solution) so this change translates to a 30% increase in ocean acidity.
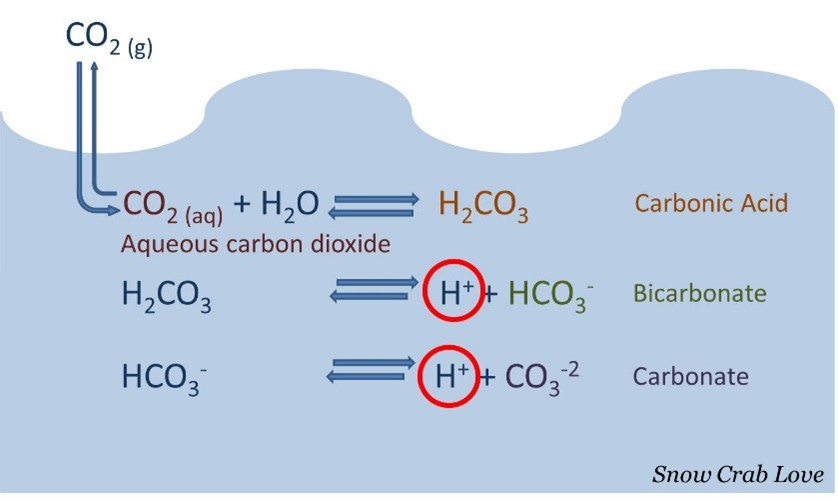
When CO2 enters the ocean, it doesn’t just float around as an independent molecule. It will combine with water, H2O, to create carbonic acid. Carbonic acid will partially dissociate in water, releasing hydrogen ions, as seen in the image below.
With more CO2 in the water, there will be more carbonic acid. Carbonic acid releases hydrogen ions which bond to carbonate, CO3. The thing is, marine organisms need that CO3 to bond with calcium in the water to form CaCO3, or calcium carbonate. Essentially the hydrogen ions that bond with the CO3 group are limiting CO3 from bonding with calcium.
When the concentration of hydrogen ions increases in the water, the pH will go down. This is concerning because organisms need calcium carbonate to build their structures and shells.
Now I want to point out that there is a place for all forms of carbon in the ocean. Carbon dioxide, carbonic acid, bicarbonate, and carbonate all have their place in a balanced ocean environment. It is the unnatural anthropogenic input of CO2 into the atmosphere that is shifting this balance and altering the ocean’s chemistry.
Now that we understand how excess carbon dioxide is harmful, we can re-visit the idea of iron fertilization.
In short, iron fertilization would work to reverse warming trends because with a massive algae bloom, tons of carbon dioxide would be taken out of the atmosphere and sequestered to the deep ocean, thereby cooling the Earth. In high nutrient, low chlorophyll (HNLC) zones, there are very little to no phytoplankton even though there are plenty of nutrients and light. The most common “explanation” is that zooplankton are decimating the phytoplankton, but in the 1930s Joseph Hart (English scientist) had a different idea. He hypothesized that these areas were limited by iron. Martin tested this theory and found that these waters were indeed low in iron. And this makes sense. Even we humans need trace metals in our diet to survive; metals are a significant micronutrient.
How does iron cause an ice age?
When the ocean sequesters carbon dioxide, there is no way for it to again reach the surface. So, if enough is taken out of the atmosphere the earth would cool, initiating an ice age. The earth actually goes through periods of cooling and heating, which we can study through the Milankovitch Cycles. But that’s another blog for another day.
The way iron fits into this theory is how it may act as a catalyst for an ice age. When water freezes, the fresh water will freeze first. So, the majority of the fresh water is frozen on earth, leaving very dry land. Winds would blow the dust off the dry land, depositing iron and other nutrients in the ocean. Nutrients would feed the algae which draw carbon into the ocean, lowering atmospheric carbon levels and continuing the cooling of the earth.
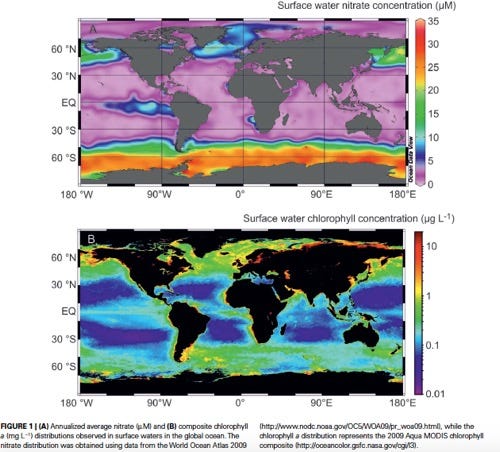
HNLC areas. Top map shows purple areas are low in nitrate. Bottom map shows dark blue areas are low in chlorpophyll.
Martin unfortunately died before he could test his iron hypothesis outside of the lab. His colleagues instead tested it in an HNLC zone off the coast of the Galapagos, where they indeed proved the hypothesis and saw an algae bloom. This means that not only are phosphorous and nitrogen vital for algae and phytoplankton growth, but metals are as well.
This theory and following experiments allowed for a larger understanding of ocean processes.
But as with any good theory, there are downsides and potential risks.
Stakeholders in the global carbon game were thrilled with Martin’s theory because that meant they could potentially feed oceans with iron to mitigate their own excessive CO2 contribution in the atmosphere. This would not only allow them to continue ‘business as usual’ in production but would put them within compliance with the Kyoto Protocol.
Well that sounds great. But what does happen when we have a massive algae bloom? There cannot be a change in one facet of the environment without initiating other changes.
When there is a massive algae bloom, we cannot control what type of algae is blooming. This means that poisonous algae could bloom and release toxic gases to the atmosphere. So, we really cannot just throw half tankers of iron in the ocean no matter how much we want to. There are tremendous benefits and knowledge gained through new findings, but the misuse of those findings is what alters the system and creates new problems that need more solutions.
What we learn from this theory and following experiments is that life in the ocean also depends on micronutrients and trace metals, and of course, that more research is needed.